HELYX QUALITY
Unified Compliance for Document Control & Training in Life Sciences
Helyx Quality
Helyx simplifies compliance by integrating document management and training into a single, cloud-based solution. By combining Helyx Controlled Documents and Helyx User Learning, it streamlines workflows, ensures regulatory alignment, and keeps teams always audit-ready.
Powered by Microsoft Azure, Helyx is pre-validated for 21 CFR Part 11, GxP, GDPR, and HIPAA, offering secure, scalable, and cost-effective quality management for biotech and pharma companies—accelerating compliance without slowing innovation.
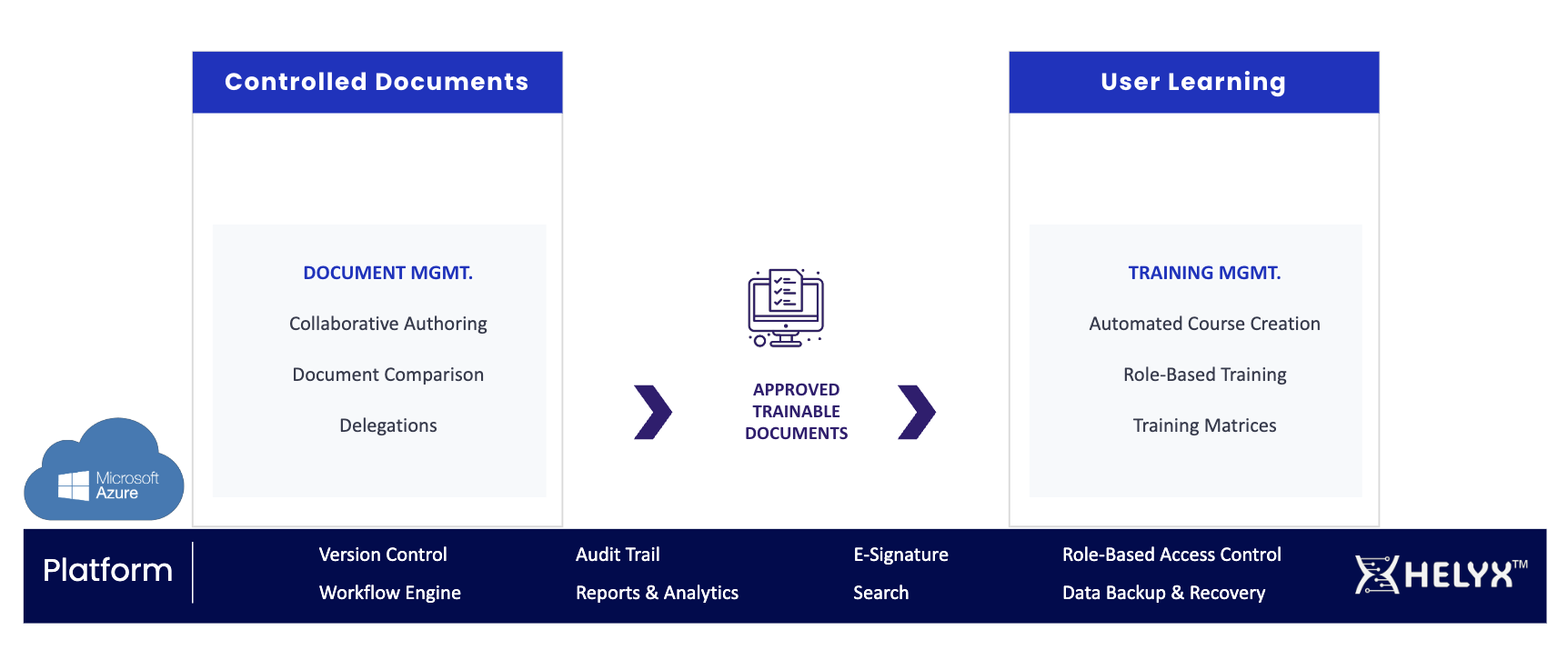
Key Modules of Helyx Quality
Helyx Controlled Documents
Helyx Controlled Documents streamlines document management while ensuring full traceability and compliance, helping life sciences companies improve efficiency and regulatory readiness. Pre-configured workflows, fast deployment, modular design and scalable pricing makes it cost-effective for early-stage and mid-sized biotechs, while seamless integration with Helyx User Learning ensures unified compliance.
Helyx User Learning
Helyx User Learning module simplifies training management with end-to-end training content creation, third-party content support, and compliance reporting. This module seamlessly integrates with the Helyx Controlled Documents Module to provide consistent compliance and easily accessible content across your organization.
